Provides an overview of ICH Climatic Zones, scheduled time points, and the key parameters customers can select to evaluate during a stability study.
Where Are Your Products Being Sold?
The lab must know where your product will be sold to design a stability protocol with the correct parameters, such as temperature and humidity. Choose from the following countries/regions:
- United States
- Europe
- China
- Canada
- Japan
- Middle East
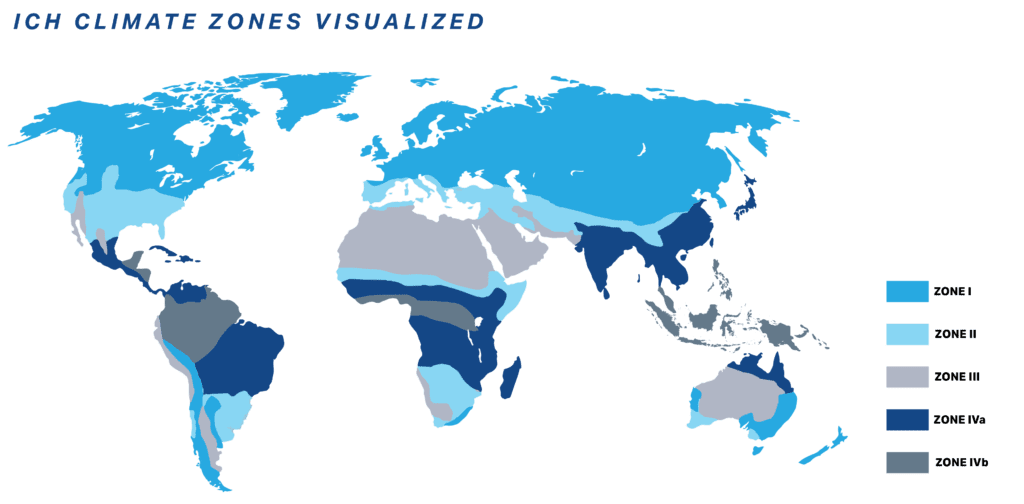
The following Zones represent different climate conditions to ensure products remain stable and effective in the regions where they’re sold.
| Zone | Description | Temperature | Relative Humidity | Geographical Areas |
| I | Temperate zone | 21°C | 45% RH | Southern Canada, Europe, Parts of Russia |
| II | Mediterranean/Subtropical Zone | 25°C | 60% RH | Mediterranean region, parts of Australia, Southern U.S. |
| III | Hot/Dry Zone | 30°C | 35% RH | North Africa, Middle East, Desert areas in the U.S. |
| IVa | Hot Humid/Tropical Zone | 30°C | 65% RH | Southeast Asia, Central Asia, Africa, Parts of South America |
| IVb | Hot/Higher Humidity Zone | 30°C | 75% RH | Regions near the equator, dense rainforest areas |
Choose the Appropriate Time Points
A time point is a scheduled stage in a stability study when a sample is pulled and tested to assess changes in quality, potency, or appearance over time. The following time points are customary for a stability study:
- Accelerated: 0, 1, 2, 3, 6 months
- Long-Term: 0, 1, 2, 3, 6, 9, 12, 18, 24, or 36 months
Test the Correct Number of Batches
It’s important to test samples from the correct number of batches to produce quality data for your study. Whether you have existing data about the batch also must be considered. Follow the guidelines below:
- Long-Term: Minimum 3 batches (if it’s a new batch and there is no existing data available).
- Accelerated: 1 batch (existing data is available).
Test Parameters Available
At each pull point, clients may select:
- Active ingredient potency (assay).
- Degradation products/impurities.
- Physical attributes (color, specific gravity, appearance, pH, odor and viscosity).
- Microbiological quality (USP <61>, <62>, <51> PET).
- Packaging compatibility (Exterior assessment ex. pump functionality, label integrity, simulated use test).
Uncertain How to Proceed? Seek Recommendations
Designing a stability study for cosmetics or OTC products requires knowledge of current regulations, industry guidelines, and scientific best practices. This can be daunting, which is why Certified Laboratories’ Stability Team provides consulting and recommendations based on our extensive knowledge of ICH, FDA, and other important guidelines and regulations. This helps you rest assured that you will receive the data you need to make the best decisions.
Ready for more? Our Cosmetics, OTC, & Supplements Resource Library contains more resources on cosmetics and OTC drug stability testing:
- Cosmetics & OTC Stability Testing Glossary
- Cosmetics & OTC Stability Testing | 101 Guide
- Cosmetics & OTC Stability Testing | 201 Guide
Questions? Contact our Stability Team!
If you need stability testing services, contact our Stability Team for help.

