 Matt Traynor, PhD
Matt Traynor, PhD
 Matt Traynor, PhD
Matt Traynor, PhD
Why 1,4-Dioxane is Difficult to Measure in Cosmetic Matrices
1,4-dioxane is a trace by-product of ethoxylation, a common chemical process used to create surfactants and emulsifiers found in many cosmetics, personal care, and OTC products.
From an analytical standpoint, 1,4-dioxane testing poses a challenge because many of these matrices are chemically “noisy”. Oils, emulsifiers, fragrances, and surfactants can generate background signals and interference that complicate identification and quantitation of 1,4 dioxane at low-ppm levels.

Why Low-PPM Levels Matter for 1,4-Dioxane Analysis
Why the need to detect this compound at low levels? And how low is “low”?
Regulations and consumer safety.
New York State regulations set a “low” bar for 1,4-dioxane detection in these product types:
- Household & Personal Care Products: ≤ 2 ppm (Dec 2022) → ≤ 1 ppm (Dec 2023).
- Cosmetics: ≤ 10 ppm (Dec 2022) (limits may be lowered further as testing technologies improve).
This approach makes sense given that 1,4-dioxane is classified as a possible human carcinogen (IARC Group 2B). As such, other states, such as California, are evaluating similar limits.

Why Traditional Methods Fall Short for 1,4-Dioxane Analysis
Fair enough, but how are manufacturers supposed to detect this compound in their raw materials or finished products given the inherent challenges of working at such low limits of detection (LOD)?
- USP <467>, a method routinely used for residual solvents, has an LOD of 380 ppm – not nearly sensitive enough for these regulatory requirements.
- EPA 8270 requires complex modifications for these matrices, which leads to inconsistent results.
Traditional methods also are run on GC-FID, which relies on retention time and column chemistry to detect 1,4-dioxane. This can cause false positives due to interfering peaks.
Here is a chromatograph from the USP <467> method:
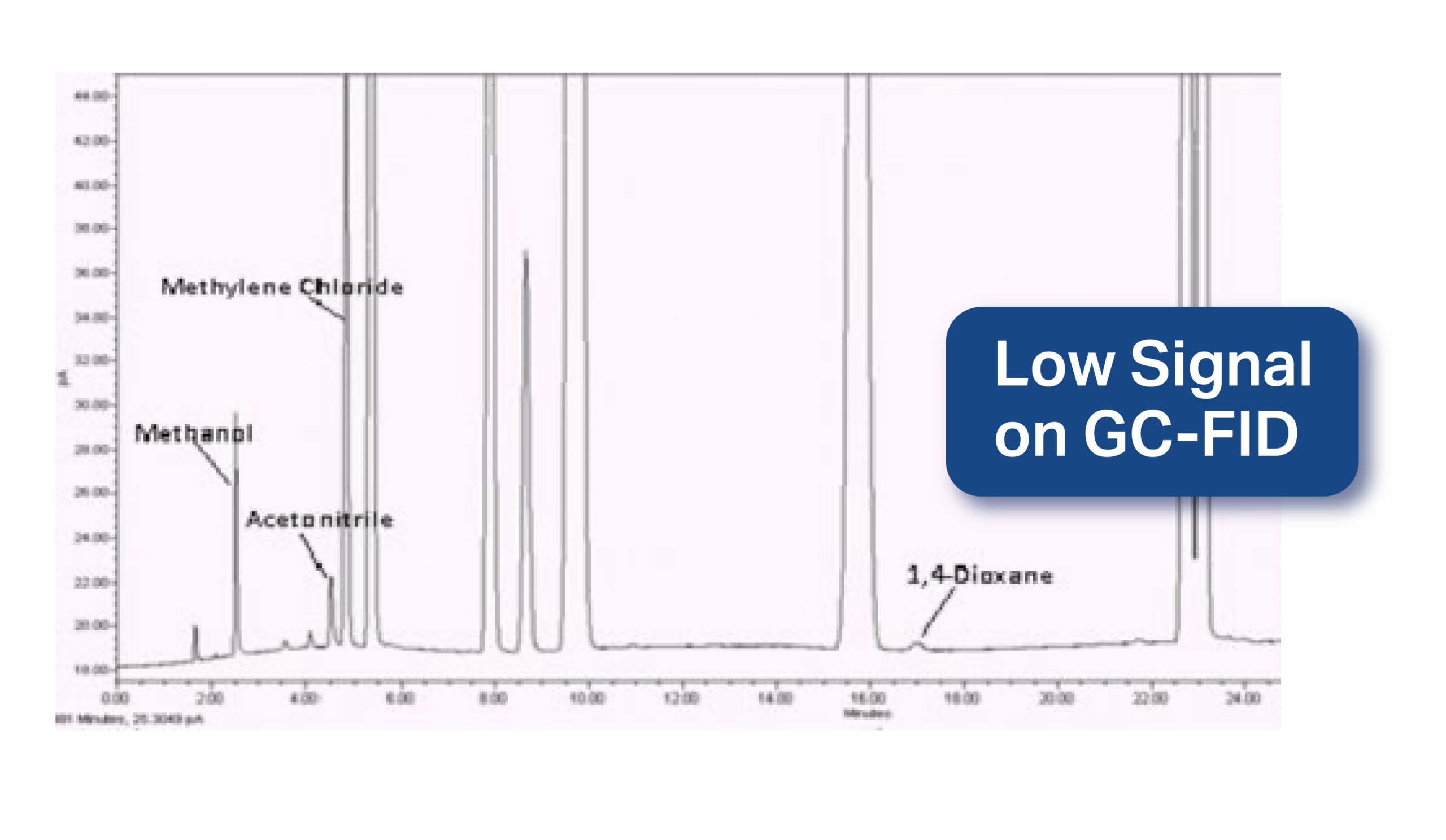
Notice how small the peak is for 1,4-dioxane and its low area count and signal. So, how do we solve this problem?
Our Approach: Validated GC-MS Method Built for Cosmetic and Personal Care Matrices
The solution is to use a method capable of detecting 1,4-dioxane down to 0.5 PPM in these matrices. Certified Laboratories’ method includes:
- Instrument Type: Single-quadrupole GC-MS configured for volatile organic compound (VOC) analysis.
- Ionization Mode: Electron Ionization (EI), optimal for small, semi-volatile contaminants like 1,4-dioxane.
- MS Scan Mode: Selected Ion Monitoring (SIM) to dramatically improve sensitivity and signal-to-noise ratio.
- Headspace Injection: Isolates the volatile solvents from non-volatile sample matrices. Prevents contamination, improves sensitivity and ensures accurate, reproducible analysis.
Why it Matters?
- SIM mode + tailored GC column = lower detection limits down to 0.5 ppm LOD, far outperforming USP <467> or general EPA methods.
- Separating out interference: Unlike GC-FID, GC-MS allows identification of compounds based on mass-to-charge ratios.
- USP method has low area counts for dioxane standard of 380 ppm.
How Does it Work?
Each compound we detect produces a unique ion signature. We compare these signatures to the spectrum of the dioxane standard. This helps us confirm if a sample really contains dioxane or just interfering compounds.
Below is the typical mass spectrum for dioxane:
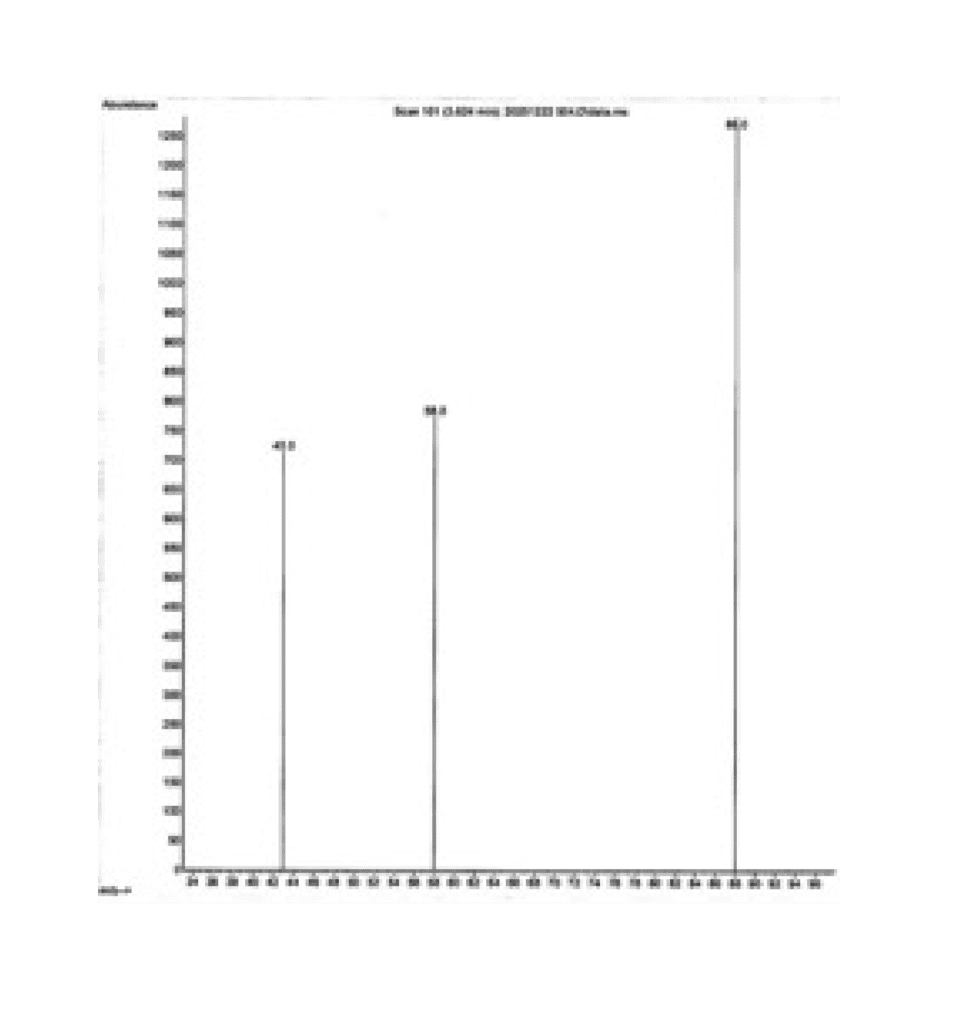
For What Products Does the GC-MS 1,4-Dioxane Analysis Method Work?
As we’ve said, cosmetics, personal care products, and OTC products can be chemically “noisy”, which makes them difficult to analyze.
To solve this problem, we designed a matrix-optimized method:
- Matrix-optimized solvent extraction to pull dioxane away from fatty or surfactant-rich environments. Tailored for both water-soluble and water-insoluble matrices.
- Water-Soluble Matrices: Shampoos, Cleansers, Hair Gels, Facial Serums
- Water-Insoluble Matrices: Lotions, Sunscreens, Lip Balms
- Low-volume injections to reduce system contamination from waxy matrices. Includes splitting mechanism to further dilute injection.
These steps ensure high recovery, low noise, and accurate quantification across creams, lotions, gels, cleansers, and surfactant systems.
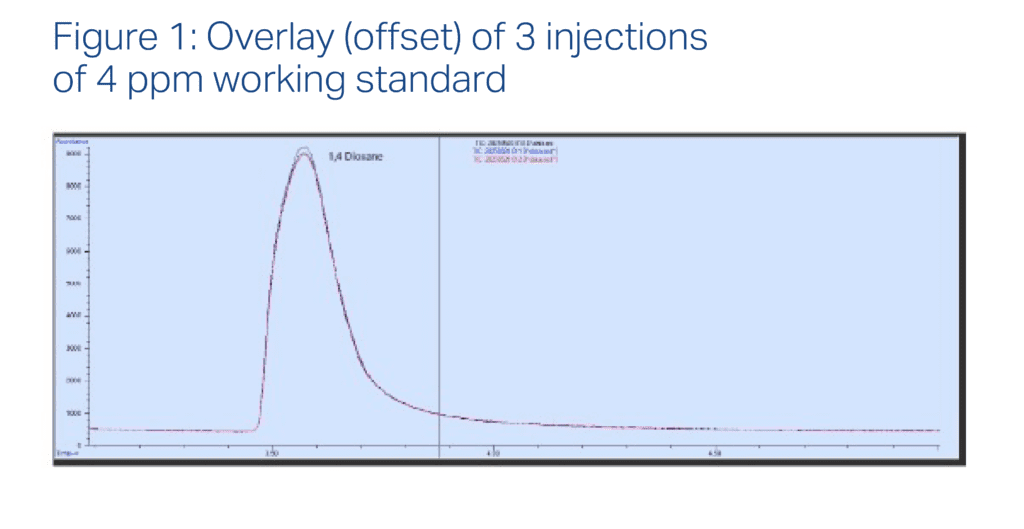
This Method is Validated – and Here’s the Data to Prove it
Let’s start by looking at the chromatogram of our 1,4-dioxane standard. Notice the clean peak and absence of noise:
And this chromatogram shows our 1,4-dioxane standard peak with retention time of ~3.5 minutes:
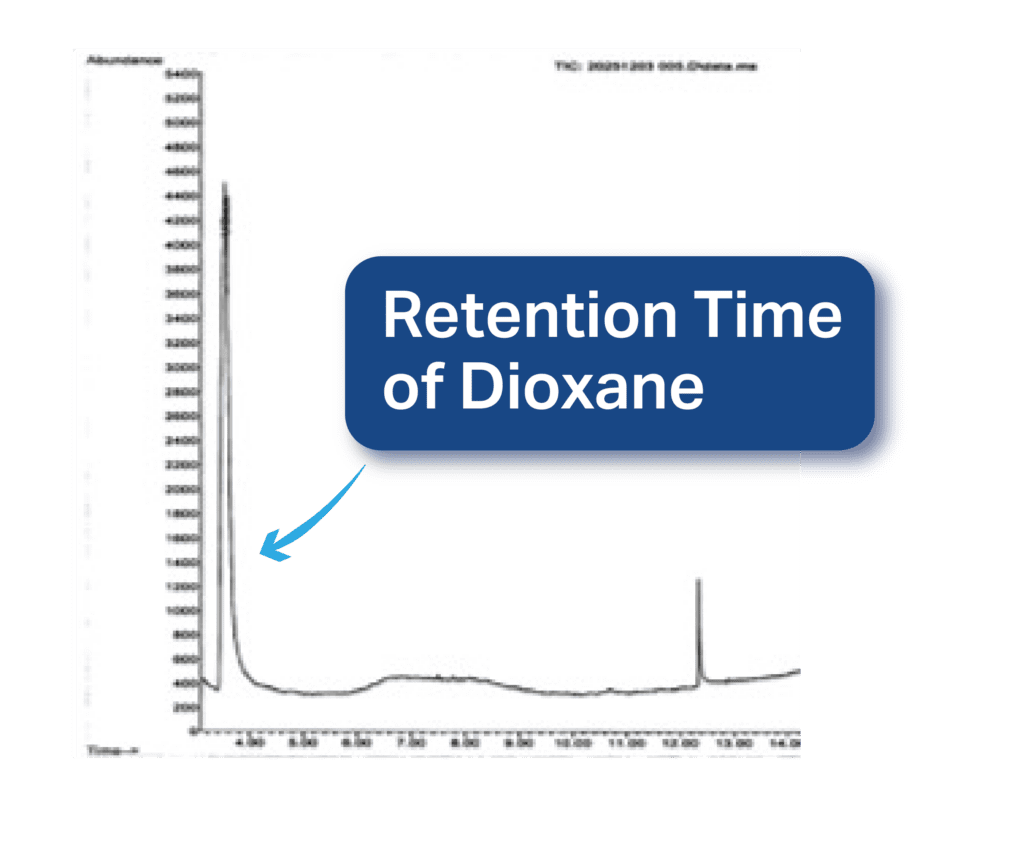
Now, look at the chromatogram from a sample that we tested in our lab:
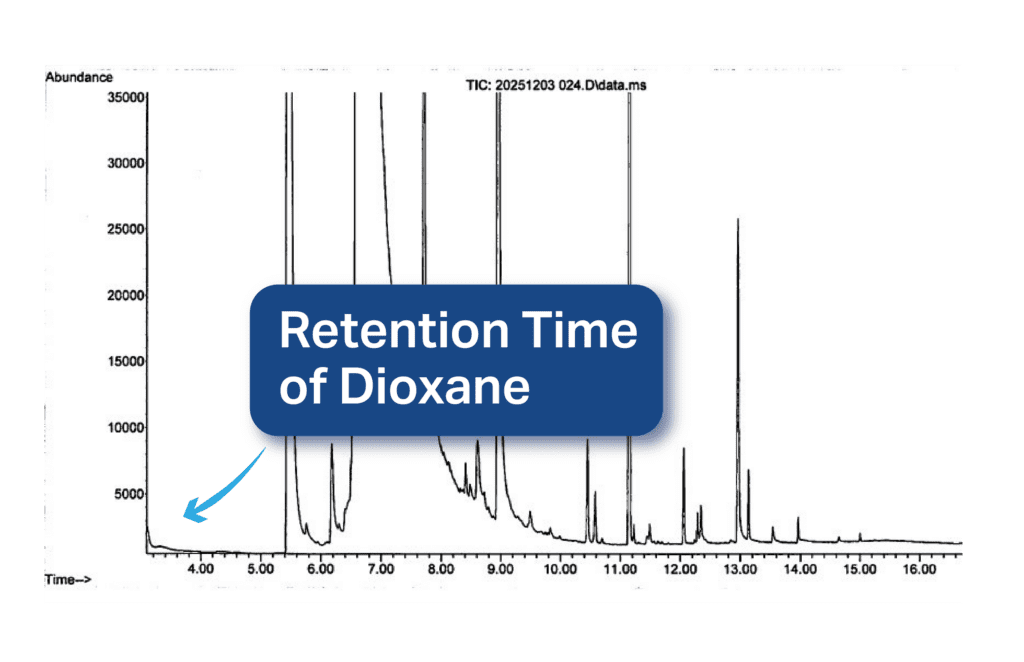
Notice that, although other compounds are detected at later retention times, at ~3.5 minutes (the retention time of dioxane), there is no peak, meaning that this sample is dioxane-free.
Next, let’s look at the linearity curve:
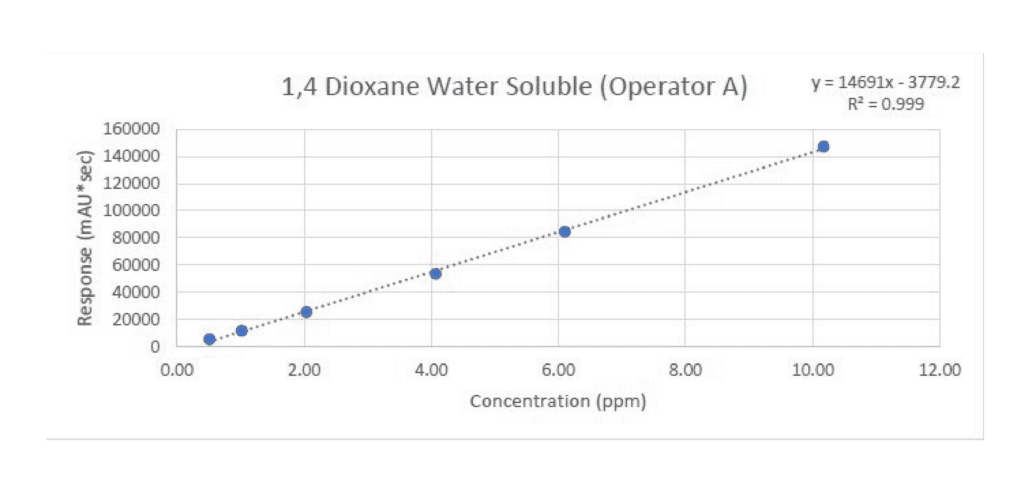
This is a plot of our dioxane standard at different concentrations versus their area counts. When we test the sample and we detect the dioxane peak, we can plot its area along this curve and quantify the concentration of dioxane that is present.
Finally, here we have data from spiking studies that we performed. Two operators prepared replicates of a spiked sample, and their recoveries had low %RSDs, confirming the precision of the analysis.
| Operator A | Operator B | |
| Experimental Concentration (ppm) | Experimental Concentration (ppm) | |
| 1.659 | 1.481 | |
| 1.647 | 1.473 | |
| 1.597 | 1.555 | |
| 1.682 | 1.560 | |
| 1.608 | 1.470 | |
| 1.617 | 1.529 | |
| Avg. Assay %RSD | 1.635 | 1.511 |
| 1.827 | 2.751 |
What Will Show Up on My COA?
After testing your samples, our cosmetic testing lab will issue a Certificate of Analysis (COA) that includes one of two outcomes:
- Not detected (which is ≤ 0.5 ppm, our limit of detection and quantification)
- If dioxane is detected above 0.5 ppm, quantity in either ppm or %, depending on your request
Here’s a sample COA:

What Makes Your Method Defensible?
Our 1,4-dioxane method includes validation components aligned with ICH Q2(R2) and ISO 17025 expectations:
- Specificity: No interfering peaks from fragrances, essential oils, silicones, preservatives, or surfactants.
- Linearity: R^2 > 0.995, LOD 0.5 ppm, standard curve from 0.5 ppm to 10 ppm.
- Accuracy: Multiple matrix spikes (low/mid/high).
- Precision: Intra- and inter-day reproducibility documented.
- Ruggedness: Analyst-to-analyst and instrument variability tested.
- Robustness: Small changes in temperature, flow, and extraction conditions do not affect quantitation.
- FULLY VALIDATED FOR COSMETIC PRODUCTS.
This translates into a method that produces defensible, reproducible results for regulatory submission, internal QA, and supplier verification.
Contact Us for 1,4-Dioxane Analysis
Our labs are ready to test your cosmetic, personal care, and OTC sample for 1,4-dioxane so you can confidently verify the safety of your products. Contact us today to discuss your testing needs.
