Reviewed By Megan Vincent, Sr. Analytical Chemist, Certified Laboratories
1-Minute Summary

- Amazon compliance testing requires using an ISO 17025-accredited laboratory for certain high-risk supplement categories.
- Weight loss, sexual wellness, and joint health products must be screened for specific undeclared pharmaceutical compounds.
- Choosing an accredited supplement testing lab with fit-for-purpose methods helps reduce delisting risk and support ongoing Amazon compliance.
- Our validated LC–MS/MS panels demonstrate linearity, precision, accuracy, and specificity for reliable presence/absence screening.
Amazon Compliance Testing for High-Risk Supplements Must Demonstrate Absence of Pharmaceutical Compounds
Sellers of supplements on Amazon continue to face evolving policy requirements. Significant updates in 2024 and again in early 2026 strengthened consumer safety standards but increased the burden on sellers, particularly in product testing and documentation.
A compliance flag can quickly lead to delisting, interrupted sales, expedited testing costs, delayed inventory release, and lost ranking momentum. As a result, Amazon compliance testing has become a core risk management function.
Amazon requires testing to be performed by ISO/IEC 17025-accredited laboratories, and certain high-risk supplement categories must demonstrate absence of specified pharmaceutical compounds. This article outlines those requirements and presents a validated approach to Amazon compliance testing designed to reduce delisting risk.
Remake graphic:

ISO/IEC 17025 Accreditation Is Foundational
First and foremost, Amazon compliance testing must be conducted by an ISO 17025-accredited laboratory.
ISO 17025 accreditation provides assurance that:
- Test methods are validated and controlled.
- Instruments are properly maintained and calibrated.
- Analysts are trained and qualified.
- Data integrity is maintained.
Accreditation signals technical competence and procedural rigor. It should be the first criterion when selecting a laboratory for Amazon compliance testing of high-risk supplements.
Amazon Compliance Requirements for High-Risk Supplements
Amazon compliance testing for certain supplement categories focuses on the absence of undeclared pharmaceutical ingredients.
High-risk categories include:
- Weight loss and weight management
- Sexual wellness
- Pain, inflammation, and joint health
The regulatory determination is binary: presence or absence of targeted active pharmaceutical ingredients. The analytical method used should be designed as a presence/absence screen with appropriate validation.
Certified Laboratories’ validated panels target the following compounds:
Weight Loss/Weight Management Panel
Stimulants and Appetite Suppressants
- 1,3-DMAA
- 1,3-DMBA
- 1,4-DMAA
- β-Methyl phenylethylamine
- Dapoxetine
- Desmethylsibutramine
- Deterenol
- Didesmethylsibutramine
- Fluoxetine
- Lorcaserin
- Octodrine
- Oxilofrine
- Phenolphthalein
- Phenpromethamine
- N, alpha-diethylphenylethylamine
Sexual Wellness Panel
PDE-5 Inhibitors and Structural Analogs
Compounds include:
- 2-Hydroxypropylnortadalafil
- Acetaminotadalafil
- Acetildenafil
- Acetylvardenafil
- Aildenafil
- Aminotadalafil
- Avanafil
- Benzamidenafil (Xanthoanthrafil)
- Benzylsildenafil
- Carbodenafil
- Chlorodenafil
- Chloropretadalafil
- Dapoxetine
- Dimethylacetildenafil
- Gendenafil
- Gisadenafil
- Homosildenafil
- Hydroxyacetildenafil
- Hydroxychlorodenafil
- Hydroxyhomosildenafil
- Hydroxythiohomosildenafil
- Hydroxythiovardenafil
- Hydroxyvardenafil
- Imidazosagatriazinone
- Lodenafil carbonate
- Mirodenafil
- N-butylnortadalafil
- N-desethylvardenafil
- N-desmethylsildenafil
- Nitrodenafil
- N-octylnortadalafil
- Noracetildenafil
- Norneosildenafil
- Norneovardenafil
- Nortadalafil
- Piperiacetildenafil (Piperidino acetildenafil)
- Propoxyphenylhomohydroxysildenafil
- Propoxyphenylsildenafil
- Propoxyphenylthiohydroxyhomosildenafil
- Pseudovardenafil
- Sildenafil
- Tadalafil
- Thioaildenafil
- Thiohomosildenafil
- Thiosildenafil
- Udenafil
- Vardenafil
- Acetaminophen
- Chlorpheniramine
- Dexamethasone
- Diclofenac
- Ibuprofen
- Indomethacin
- Meloxicam
- Methocarbamol
- Naproxen
- Omeprazole
- Piroxicam
- Prednisolone
- Prednisone
Inclusion of structural variants is critical. Modified analogs may be introduced to evade limited screening approaches. Comprehensive coverage strengthens compliance assurance.
Joint Health/Pain & Inflammation Panel
NSAIDs, Corticosteroids, and Related Compounds
- Diclofenac
- Ibuprofen
- Naproxen
- Indomethacin
- Meloxicam
- Piroxicam
- Methocarbamol
- Acetaminophen
- Prednisone
- Prednisolone
- Dexamethasone
- Hydrocortisone
- Chlorpheniramine
- Omeprazole
Detection of any listed compound constitutes non-compliance under the screening requirement.
A Validated Approach to Amazon Compliance Testing
To support sellers in high-risk categories, we developed panels using LC–MS/MS with electrospray ionization and multi-point calibration. The analytical platform is standardized across categories to promote consistency and efficiency.
Our validation process includes:
- Linearity: Calibration curves demonstrated correlation coefficients of 0.99 or greater across defined concentration ranges. This confirms predictable instrument response as concentration varies.
- Precision: Replicate injections produced relative standard deviations below 5%, demonstrating repeatability and minimizing analytical variability.
- Accuracy and Recovery: Spike recovery studies were conducted by fortifying representative supplement matrices with known analyte concentrations. Recoveries fell within ± 15 percent of expected values, confirming reliable detection in complex product matrices.
- Specificity: Blank matrix samples showed no interfering peaks at target retention times, reducing the likelihood of false positive signals.
Collectively, these validation elements demonstrate that the method is suitable for Amazon compliance testing in the specified categories.

Interpreting Data in Amazon Compliance Testing
Let’s look at a few representative chromatograms to show the validation work of these methods.
Joint Health/Pain & Inflammation – Acetaminophen
- r^2: 0.993
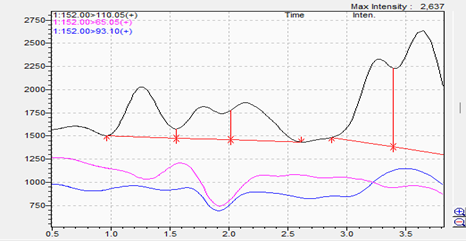
Blank:
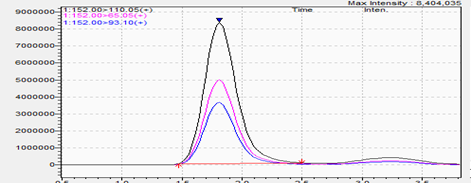
10 ppm Standard:
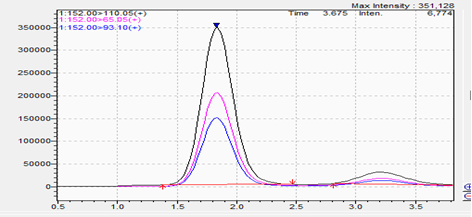
Spike:
Weight-Loss/Weight Management: 1,3 DMAA
- r^2: 0.995
Here are more for analysis of 1,3 DMAA, which is prohibited in weight-loss supplements.
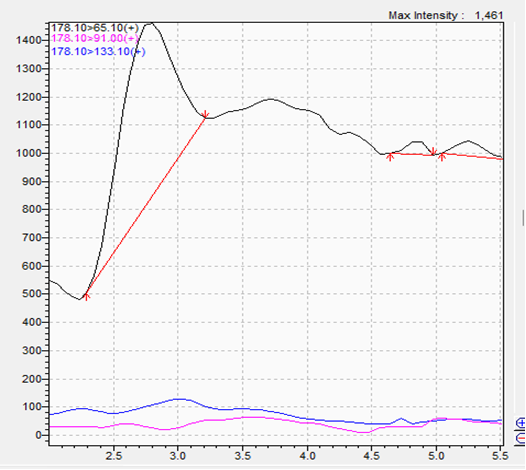
Blank:
10 ppm Standard:
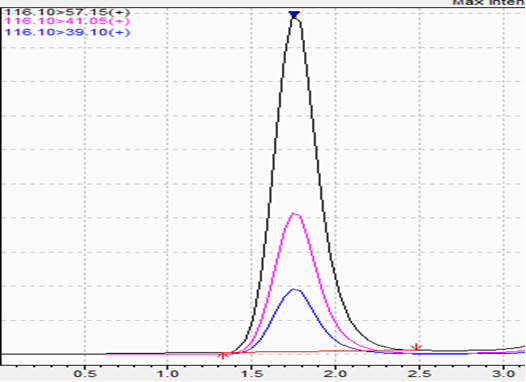
Spike:
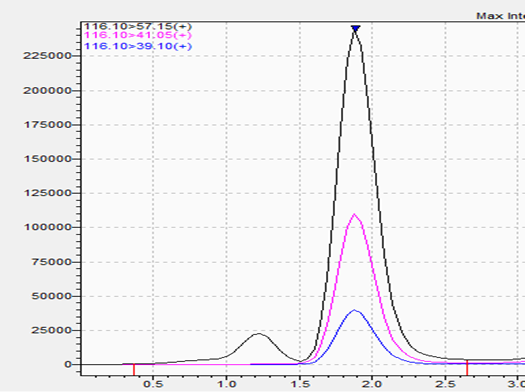
What Do These Chromatograms Mean?
- The blank samples demonstrate absence of interfering peaks in the product matrix.
- Low-level standards establish reference signals for targeted compounds at defined ppm levels.
- Matrix spikes confirm that targeted analytes are detectable in real supplement formulations and that signal characteristics align with validated expectations.
These data illustrate that the method detects targeted compounds when present and does not generate misleading signals when absent. This supports confidence in Amazon compliance testing results.
Strengthen Your Amazon Compliance Strategy
If you sell any of these high-risk supplements on Amazon, make validated testing at an ISO 17025-accredited laboratory a key part of your process. Doing so helps ensure product safety and compliance with Amazon’s supplement policy.
Our Joint Health and Weight Loss Panels have completed validation. Our Sexual Wellness panel validation is in progress. Planned future Amazon compliance testing panels include:
- Sports Performance Products & DHEA
- Blood Glucose Management & Support Products
- Nootropic & Sleep Support Products
- Cardiovascular & Hypolipidemic Support Products
Certified Laboratories is an Amazon-approved Testing, Inspection, and Certification (TIC) organization for supplements, so you can trust our results. Contact our ISO 17025-accredited supplement testing laboratories to discuss your Amazon compliance testing needs and reduce delisting risk.

