 Matt Traynor, PhD
Matt Traynor, PhD
 Matt Traynor, PhD
Matt Traynor, PhD
Uncertainty creates stress. It slows decision-making, increases risk, and makes the job of safety and quality professionals more difficult.
One common source of that stress is not knowing whether a supplement or OTC drug sample needs feasibility, method validation, or method verification. Is the method appropriate for your matrix or formulation? What is each one, and when is it needed for regulatory purposes?
We created a webinar to help answer those questions. If you need answers now, this FAQ gathers the top questions to help your team move toward a clearer, more defensible testing program – and reduce some of the stress that comes with uncertainty.
1. What is the difference between method feasibility, method validation, and method verification?
- Feasibility: Tests a new or unique matrix that has never been tested before. It assesses if the analyte can be properly extracted, detected, and measured in the product matrix.
- Method Validation: Confirms a testing method is accurate, consistent, and reliable for a specific analyte, ensuring the target ingredient can be measured accurately without interference. Validation is performed once per test method.
- Method Verification: Confirms an existing validated method performs as expected for a specific formulation, sample type, or matrix. It ensures the method remains suitable and produces consistent results across product variations.
2. How do you determine whether a product needs feasibility, validation, or verification testing?
The answer depends on what is new.
Use feasibility when the product introduces a new or untested matrix and the main question is whether the existing method will work with it. This often applies to supplements such as gummies, botanicals, soft gels, or tablets.
Use validation when the method itself is new. For example, if a new sunscreen UV filter has no existing validated method, the method must be validated.
Use verification when a validated method already exists, but it must be shown to work for a specific formula or matrix. If a verified product is later reformulated, it typically needs verification again.
The table below provides examples:
| New Dietary Supplement Analyte or Ingredient > Feasibility |
| Unique or Complex Supplement Matrix > Feasibility (e.g., gummies, botanicals, softgels) |
| New SPF Matrix with No Applicable Validated Method > Method Validation |
| New SPF Formulation > Method Verification (different format/matrix) |
| Reformulation or Line Extension > Method Verification (same actives, new base) |
| New Active with No Existing Method > Method Development and Validation |
| Raw Material or Finished Goods where Existing Compendial Method is Available > Run Compendial Method Verification |
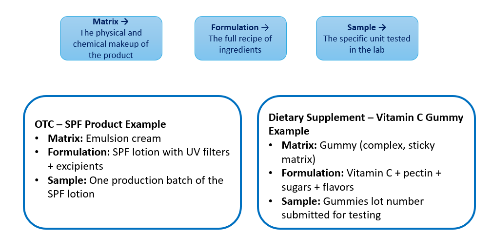
3. What do matrix, formulation, and sample mean, and why do those distinctions matter for analytical testing?
Matrix is the product’s physical and chemical form, such as a gummy, powder, tablet, cream, or lotion.
Formulation is the full recipe of ingredients.
Sample is the specific batch or unit sent to the lab.
These differences matter because the method has to work in the actual product, not just on paper. An SPF lotion in an emulsion cream matrix behaves differently from a vitamin C gummy made with pectin, sugars, and flavors, so extraction and suitability can change.
4. When does a reformulation, line extension, or matrix change trigger a new method verification?
A new verification is usually needed when a product is reformulated, extended into a new line, or moved into a new matrix. Those changes can affect whether the method still measures the analyte accurately and consistently.
Not every variation requires a full repeat. For products with multiple shades or flavors built on the same base formula, it may be enough to verify the worst-case version, such as the darkest shade or strongest flavor, and assess the others through specificity work.
5. What regulatory expectations apply to method validation and verification for OTC drug and dietary supplement products?
For OTC drugs, 21 CFR 211 requires test methods to be established, documented, and shown to be suitable under actual conditions of use.
For dietary supplements, 21 CFR 111.320 requires firms to use scientifically valid methods and verify that testing methods are appropriate for their intended use.
The main frameworks referenced are ICH Q2, FDA guidance, USP <1225>, and ISO 17025. The practical expectation is clear: firms need documented evidence that the methods supporting label claims, release, and stability work are valid for the product being tested.
6. What data and criteria are typically included in method validation, verification, and feasibility reports?
A feasibility report is relatively short. It typically includes the method used, acceptance criteria, results from three replicates, relative standard deviation, and any limitations observed.

Validation and verification reports are more detailed. They may include method description, instrument details, acceptance criteria, summary data, statistical analysis, measurement of uncertainty, and conclusion.
Common criteria include…
- Specificity
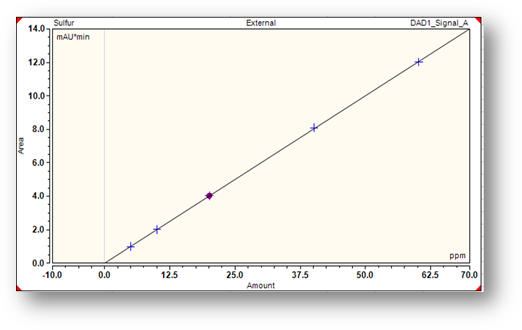
- Linearity
- LOD
- LOQ
- Accuracy
- Precision
- Intermediate precision
- Robustness
Verification focuses more narrowly on product-specific performance, including forced degradation when applicable.
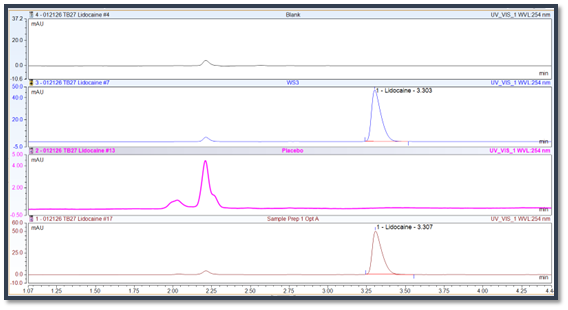
7. What should a company do if a method verification or validation fails for a specific product or formulation?
When that happens, the method usually needs development work so the analyte can be separated and measured correctly.
A failed validation or verification usually means the current method is not suitable for that product. One common example is interference from another peak that co-elutes with the active and prevents accurate quantitation.
Compendial methods also have limits. If a USP method is changed beyond what the monograph allows, it stops being a USP method and becomes an in-house method that must be validated accordingly.
Why this Matters to Your Brand
Method validation, verification, and feasibility all serve the same purpose: helping your team build a stronger, more defensible testing program.
They support reliable data, reduce risk, improve documentation, and confirm that your methods are appropriate for the products you make so you can move forward with greater confidence in quality, compliance, and consumer trust.
Contact Certified Laboratories if you have questions about what you need for your testing program.
