This guide covers the critical steps of microbiology testing, common test methods, and the importance of microbial suitability.
Primary Stages When Cosmetic Microbiology Testing Is Performed
Microbiology testing is performed at multiple stages to help ensure quality and safety:
Water: Whether used in the formulation or manufacturing process, it is one of the most common sources of contamination in cosmetics.
Raw Ingredients: Especially botanical or natural materials.
Bulk/In-Process: During mixing or compounding phases.
Finished Product: Before it’s released to market.
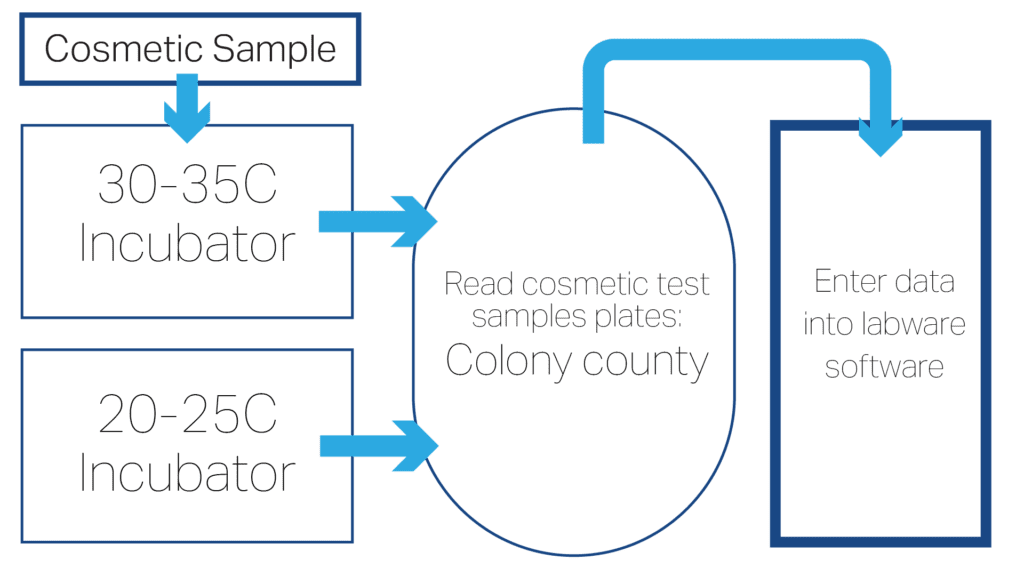
Key Micro Testing Steps
- Receive samples in the lab using aseptic techniques to ensure sample integrity.
- Neutralize product in nutrient broth.
- Transfer into petri dishes and add agars.
- Incubate: 30° to 35°C (3 days) – TAPC.
- Incubate: 20° to 25°C (5 to 7 days) – TYMC.
- Perform enrichment step.
- Read plates: using colony counter with light source and magnifying glass, look for bacteria growth on tryptic soy agar (TSA) plates. Look for yeast/mold growth on potato dextrose agar (PDA)/(SDA) plates.
- Following Standard Operating Procedure (SOP), enumerate any microbial growth and characterize any potentially pathogenic microorganisms (e.g. Staphylococcus aureus).
- Enter data into the LIMS systems.
- Review, release Certificate of Analysis (COA) (summary of data) to customer
Micro – Common Tests
- Pathogen Screening (e.g., Staphylococcus aureus, Pseudomonas aeruginosa, E. coli, Candida albicans, BTGN [quantitative/qualitative], Salmonella, Clostridium (upon request))
- Burkholderia cepacia complex (BCC) (USP 60)
- Standards Referenced: USP 60/61/62, ISO 17516
- Total Aerobic Microbial Count (TAMC or (APC)
- Total Yeast and Mold Count (TYMC or Y/M)
Microbial Suitability vs. Routine Testing
Microbial Suitability Testing
- Purpose: Proves the test method works with your product
- Validates the method for your specific formula
- Confirms microbes can be recovered from the product
- Ensures preservatives or ingredients don’t interfere with detection
- Typically performed once per new formulation
Key takeaway: Proves the test works
Routine Microbiological Testing
- Purpose: Checks if microbes are present
- Evaluates the product itself
- Measures microbial counts or detects specified organisms
- Confirms compliance with microbial limits
- Performed on production batches as needed
Key takeaway: Tests the product
The importance of micro suitability
Ensures Method Validity
Needed for any new product or formulation (typically required once).
Adding an active, fragrance, or color makes a formula “new,” as these changes can impact microbial growth and preservative performance.
Suitability testing verifies that the microbiological method can accurately detect microbes in your specific product.
Many cosmetic or OTC formulations contain preservatives, high salt or sugar levels, or other ingredients that can inhibit microbial recovery. If recovery is not verified, a “clean” result may not reflect the true microbial risk.
Regulatory Requirement
USP <61/62> (microbial enumeration and qualitative tests for specified organisms) and USP 60 (Burkholderia cepacia complex) require suitability testing as part of compliance.
Ready for more? Our Cosmetics, OTC, & Supplements Resource Library contains more resources on cosmetics and OTC drug stability testing:
- Cosmetics and OTC Microbiology | Glossary
- Cosmetics and OTC Microbiology | 101
- Cosmetics and OTC Microbiology | 201

