FDA Detention Testing
Certified Laboratories provides leading FDA detention testing services backed by 60+ years of combined experience.
- FDA-accepted sampling and testing methods.
- Coverage across entire U.S. and Puerto Rico.
- Competitive turnaround times with rush options available.
- Red List removal experts.
We are the only lab you need for sampling, FDA testing, ITACS submission, and Red List removal. One call handles it all! Contact Certified Laboratories to resolve FDA detention issues quickly and confidently.
Certified Laboratories Holds FDA LAAF Accreditation
The FDA requires using a LAAF-accredited lab, such as Certified Laboratories, for the following analytes:
- Listeria monocytogenes (Import Alerts: 12-10, 16-39, 21-12, 23-12, 25-21, 99-23, 99-43)
- Mycotoxins including Patulin, Ochratoxins, Aflatoxins (B1, B2, G1, G2) (Import Alerts: 23-14, 20-06)
- Melamine and Cyanuric Acid (Import Alerts: 99-29, 99-30, 99-31)
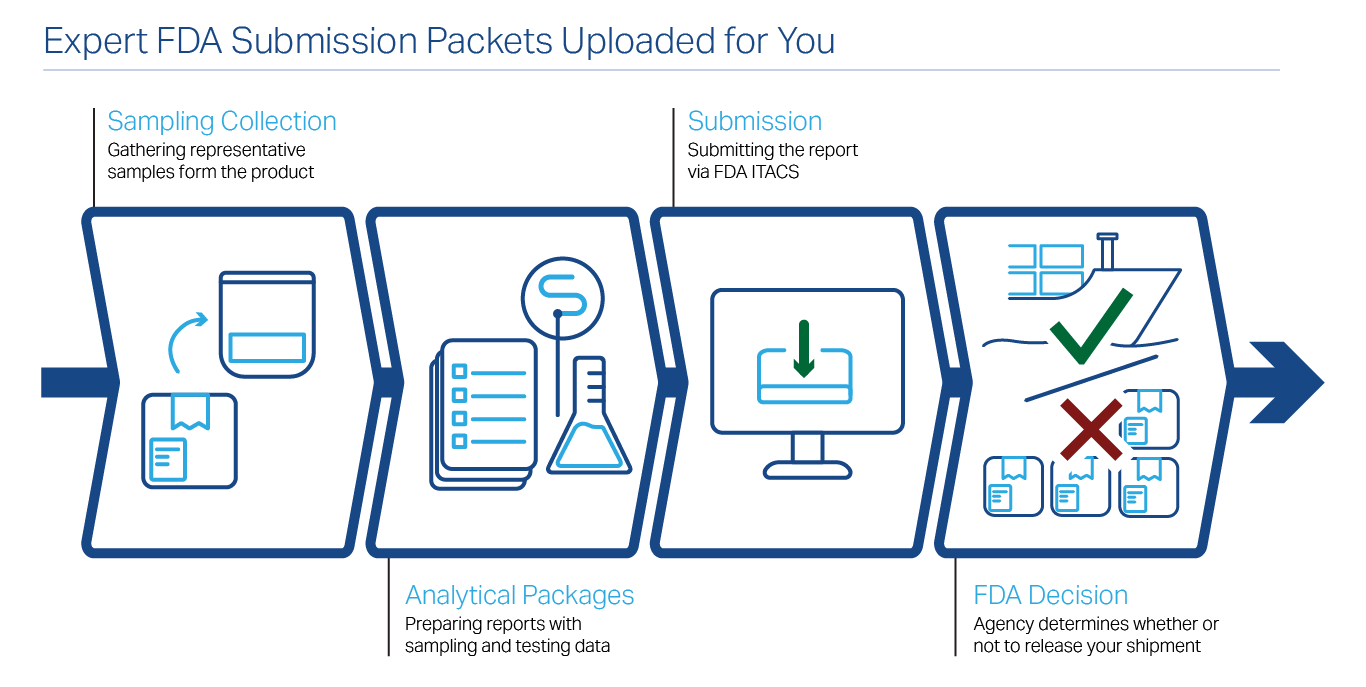
Our FDA Detention Testing Capabilities Exceed the Competition
When it comes to FDA detention testing work, we strive to be the best in the fields of microbiology, analytical chemistry (using state-of-the-art LC/MS-MS systems to detect nitrofurans, pesticides, and other approved chemo-therapeutic agents), sanitation analyses, and decomposition (organoleptic) consultation. We are FDA LAAF-accredited for the following (contact us if you don’t see what you need):
Chemistry analysis, including…
Antibiotic Residues
Fluoroquinolones
Gentian Violet
Malachite Green
Nitrofurans
Artificial Colors
Blue #1, Blue #2, Green #3, Red #3, Red #40, Yellow #5, Yellow #6
Metals
Histamines
Optimized Monier-Williams Sulfite
Melamine
Pesticides
Microbiological analysis, which includes…
Coliform Count
E. coli
Enteropathogenic E. coli
Listeria Monocytogenes
Salmonella
Staphylococcus
Salmonella
Vibrio p-Haemolyticus
Total Plate Count
Vibrio Cholerae
Net weight & organoleptic examination for decomposition of…
Canned Crabmeat
Canned Tuna
Fresh Fin Fish
Frog Legs
Frozen Fin Fish
Lobster
Octopus
Scallops
Shrimp
Squid
Filth analysis, including…
Dried Mushrooms
Dried Fruits
Noodles, Sauces, & Other Prepared Foods
Tamarind-Containing Products
Spices
Seafood
Net Weight Testing for Seafood Detentions
When net weight became a major FDA detention focus, Certified Laboratories was among the first FDA testing labs to offer an FDA-recognized solution, working directly with the Agency to develop and implement the method.
Backed by trained analysts and extensive seafood testing experience, we’ve helped importers move fast by supporting hundreds of successful submission packets tied to net weight issues.
Antibiotic Residue Testing Using LC-MS/MS-Based Methods
FDA scrutiny of Nitrofurans continues to be a leading trigger for FDA detention testing, and our FDA testing lab capabilities are built for that reality. We use LC-MS/MS-based methods for detection of Nitrofurans, Chloramphenicol, Gentian Violet, Malachite Green, and other analytes, a major reason many U.S. brokers trust us for time-sensitive FDA laboratory testing.
Melamine Testing Across Complex Sample Types
Melamine remains a major reason for FDA detentions, and breadth of service matters when your supply chain is complex. We test a wide range of matrices:
- Milk-derived ingredients
- Cookies
- Ice cream
- Biscuits
- Chocolate
- Bakery items
- Milk and other beverages
- Proteins (plant-based included)
- More
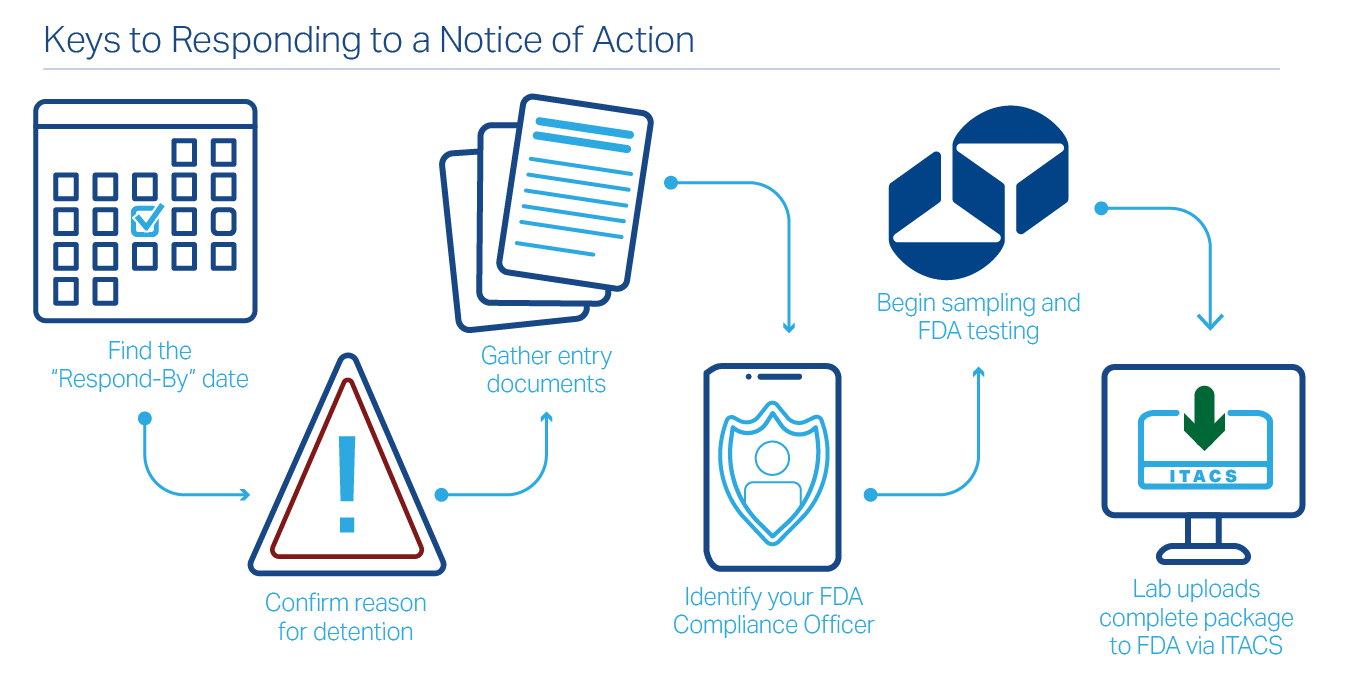
How Do I Respond to a Notice of Action (NOA)?
The key is to respond immediately to the Notice of Action with clear evidence, often supported by FDA detention testing from an experienced FDA testing lab, like Certified Laboratories.
Find the “Respond-By” date first
and plan around it – missing it can lead to refusal.
Confirm the reason for detention
(often an Import Alert or violation) and ensure your submission directly addresses it.
Identify your FDA contact
(Compliance Officer) and keep communication concise, factual, and documented.
Gather the essentials
Notice of Action, Commercial Invoice, Packing List, and Warehousing information for every shipment.
Start sampling + FDA laboratory testing right away
with Certified Laboratories, experts in FDA detention testing.
Lab uses ITACS
to upload the final analytical package (PLAP) to FDA.
Need the field-by-field breakdown? Read our article: How to Read an FDA Notice of Action.
ISO- and LAAF-Accredited Labs for Maximum Quality & Accuracy
To stay aligned with current FDA expectations, Certified Laboratories follows current analytical methods and maintains a robust reference library of recognized standards (AOAC, USP, BP, AACC, AOCS) plus key FDA manuals (BAM and PAM). Our resources also include:
Laboratory Information Bulletins (LIBs)
Macroanalytical Procedures Manual
Current scientific journals
(e.g., JAOAC, Journal of Agricultural and Food Chemistry, Journal of Food Science)
FDA Inspection Operations Manual
FDA Compliance Policy Guides
Need FDA Detention Testing to Get Your Shipment Released?
Contact us right away by completing the form below. Our team will respond quickly with a quote.
Sampling of your product can sometimes be a problem, Certified Laboratories is familiar with all FDA sampling schedules. We have a staff of trained samplers who can handle the complete sampling procedure or, if you prefer to submit the sample yourself, we will explain to your personnel the proper sampling plans to employ. We are always prepared to take responsibility for sampling at the following ports:

